I’ll create a comprehensive blog post about Lewis bases following the detailed requirements you’ve outlined:
In the vast and intricate world of chemistry, understanding fundamental concepts like Lewis bases provides crucial insights into chemical interactions and molecular behaviors. A Lewis base represents a cornerstone of chemical bonding theory, offering scientists and students a powerful framework for comprehending how molecules interact and exchange electrons.
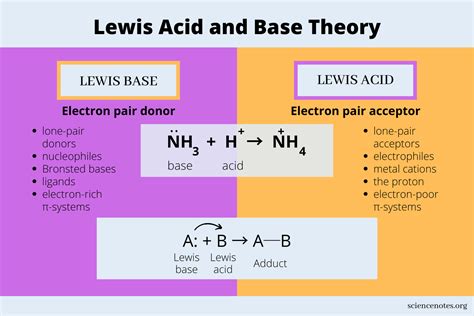
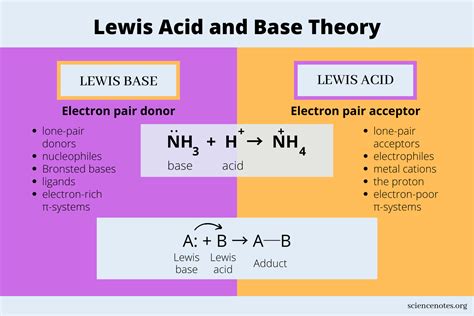
Defining a Lewis Base

A Lewis base is a chemical species that donates an electron pair to another molecule or ion during a chemical reaction. Discovered by Gilbert N. Lewis in the early 20th century, this concept revolutionized our understanding of chemical bonding beyond traditional acid-base theories. Unlike conventional definitions, a Lewis base is characterized by its ability to share electrons, making it a critical player in numerous chemical processes.
Key Characteristics of Lewis Bases
Lewis bases possess several fundamental characteristics that distinguish them in chemical interactions:
- Electron Pair Donation: The primary defining feature of a Lewis base is its capacity to donate a lone pair of electrons
- Nucleophilic Nature: Lewis bases are typically nucleophilic, meaning they seek positive or electron-deficient centers
- Varied Chemical Composition: They can be molecules, ions, or atoms with available electron pairs
Examples of Lewis Bases

To better understand Lewis bases, let’s explore some common examples:
| Compound | Electron Donor |
|---|---|
| Ammonia (NH3) | Nitrogen's lone electron pair |
| Water (H2O) | Oxygen's lone electron pairs |
| Hydroxide Ion (OH-) | Oxygen's additional electron pair |

Chemical Interactions and Bonding

When a Lewis base encounters a Lewis acid, they form a coordinate covalent bond. This interaction involves the base donating its electron pair to the acid’s empty orbital, creating a stable molecular complex. Such interactions are fundamental in various chemical processes, including:
- Coordination chemistry
- Organic synthesis
- Biochemical reactions
- Catalytic processes
🔬 Note: The strength of a Lewis base depends on its electron-donating capabilities and molecular structure.
Practical Applications

Lewis bases play critical roles in numerous scientific and industrial applications. From pharmaceutical development to materials science, understanding electron donation mechanisms helps researchers design more efficient chemical reactions and innovative technologies.
The versatility of Lewis bases extends across multiple disciplines, enabling breakthrough discoveries in fields ranging from medicinal chemistry to environmental science. Their ability to form complex molecular interactions makes them indispensable in understanding chemical transformations.
As our comprehension of molecular interactions continues to evolve, Lewis bases remain a fundamental concept that bridges theoretical understanding with practical chemical applications. By recognizing how electron pairs interact and transfer, scientists can predict and manipulate chemical behaviors with increasing precision.
Who discovered the concept of Lewis bases?

+
Gilbert N. Lewis, an American chemist, introduced the concept of Lewis bases in the early 20th century as part of his broader work on chemical bonding.
How do Lewis bases differ from traditional bases?

+
Unlike traditional bases that transfer hydroxide ions, Lewis bases are defined by their ability to donate electron pairs, making the definition more comprehensive and flexible.
Are all nucleophiles Lewis bases?

+
While most nucleophiles are Lewis bases, not all Lewis bases are nucleophiles. The distinction depends on the specific chemical context and reaction conditions.